Description
Hepatitis C virus (HCV) is an enveloped, single-stranded positive-sense RNA (9.5 kb) virus belonging to the family of Flaviviridae. Six major genotypes and a series of subtypes of HCV have been identified. Isolated in 1989, HCV is now recognized as the major cause of transfusion-associated non-A, non-B hepatitis. The disease is characterized by acute and chronic forms although more than 50% of the infected individuals develop severe, life-threatening chronic hepatitis with liver cirrhosis and hepatocellular carcinomas. Serological evidence of HCV infection may be obtained by testing for HCV antigens or antibodies in the serum of individuals suspected of hepatitis C infection. Antibodies to HCV can be detected throughout virtually the total infection period. Therefore, the use of highly sensitive antibody assays is the primary approach in serodiagnosis of HCV infection.
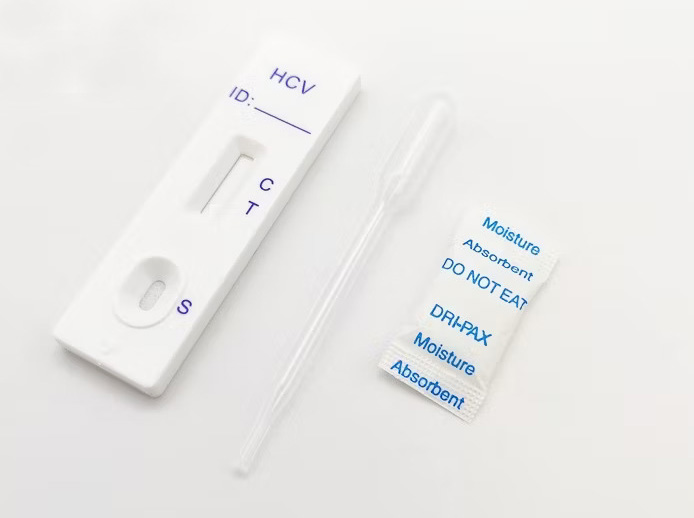
PRINCIPLE
This test is based on the immunochromatographic principle. The test device consists of a sample window containing a conjugate pad. The conjugate pad is held in contact with the porous membrane material. The membrane has three zones. The first zone is mobilized by the sample and it consists of colored colloidal gold particles coupled with HCV recombinant antigen.
The second zone consists of recombinant HCV antigens immobilized on the membrane (Test line). The recombinant HCV antigens used in this test include both structural (nucleocapsid) and non-structural proteins including NS-3, NS-4 and NS-5. The third zone (Control line) consists of control antibodies, which is also immobilized on the membrane. If an HCV antibody is present in the test sample, it will form a complex with the HCV recombinant antigen coupled conjugate and then move on, to be trapped by the test line, causing the formation of the red line. The unbound colloidal gold particles continue to move along the strip by capillary action until they come in contact with the control line and re re-rapped, giving a red line demonstrating the validity of the tests.




Reviews
There are no reviews yet.